- This page was created by volunteers like you!
- Help us make it even better. To learn more about contributing to MEpedia, click here.
- Join the movement
- Visit #MEAction to find support or take action. Donate today to help us improve and expand this project.
- Congratulations!
- MEpedia has got over 30 million views as of August 2022!
Pregnancy
Research on outcomes of pregnancy for women with ME is limited. There is some evidence of symptom improvement or remission in various inflammatory and autoimmune diseases during pregnancy. This may be due to hormonal or immune changes.[1]
Biological changes during pregnancy[edit | edit source]
Development of the placenta[edit | edit source]
The placenta is a vascular organ that develops during pregnancy, attaching to the wall of the uterus. The umbilical cord connects the placenta to the developing fetus, allowing for blood to pass from the mother. The placenta allows for nutrient supply, thermoregulation, waste elimination, and gas exchange for the developing fetus, as well as production of hormones that support pregnancy for the mother. Towards the end of the pregnancy, the placenta provides antibodies to the fetus for protection against infectious pathogens after birth.[2]
The fetus and the placenta may be detected as foreign threat by the mother's immune system. To prevent this from happening, the placenta secretes molecules containing Neurokinin-B. This is the same mechanism that parasitic nematodes use to evade detection by the host immune system. Additionally, there is a presence of lymphocytic suppressor cells in the fetus, which can inhibit maternal cytotoxic T cells.[2][3]
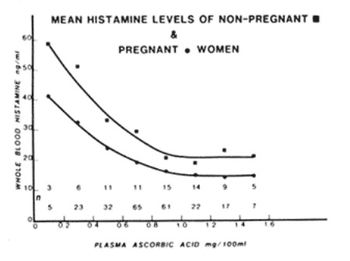
Immune system[edit | edit source]
As a prerequisite for a successful pregnancy, the immune system undergoes changes so that the mother's body will not reject the fetus.[4] To do so, there is a general shift of the immune system from the a pro-inflammatory state (Th1) to an anti-inflammatory state (Th2) during pregnancy. Helper T cells (Th) of the immune system are divided into two main categories, Th1 and Th2, that have opposite functional activity. An immune response can be categorized as a Th1 response (pro-inflammatory, usually in response to pathogens or injury) or a Th2 response (anti-inflammatory, can be used to down-regulate the Th1 state). While both Th1 and Th2 responses are at play, the immune system tends to be dominated by one or the other depending on the Th1/Th2 balance.[5]
One study has shown that T cells from pregnant women produce less Th1 cytokines and more Th2 cytokines, indicating a general shift toward the Th2 anti-inflammatory state. Additionally, levels of regulatory T cells (Tregs), progesterone, and estrogen increase during pregnancy, which have anti-inflammatory properties.[6]
In contrast, the T cells in women with recurrent spontaneous abortions produce normal, non-pregnancy levels of Th1 and Th2 cytokines, indicating a Th1 pro-inflammatory state in unsuccessful pregnancies.[4] In this way, many spontaneous abortions are similar to organ transplant rejections; a Th2 state is required for successful pregnancies and organ transplants, and failed organ transplants usually occur in people who remain in a Th1 dominant state.[4]
Pregnancy and ME/CFS[edit | edit source]
Fertility[edit | edit source]
Women with chronic fatigue syndrome report higher rates of polycystic ovarian syndrome (PCOS) and anovulatory cycles, higher rates of dysmenorrhea and higher rates of endometriosis.[7]
Symptoms during pregnancy[edit | edit source]
There has been very little research on pregnancy and ME/CFS. One retrospective survey found that 41% of respondents reported no change in symptoms during pregnancy, 30% improved and 39% got worse.[8]
In clinical practice, Dr. Nancy Klimas, Dr. Lucinda Bateman, and Dr. Charles Lapp report higher rates of improvement or remission during pregnancy. Klimas reports that in the 20 women she has followed through pregnancy, improvement in symptoms during pregnancy was "almost universal" and in some cases resulted in total temporary remission. Dr. Lapp reported that 25 out of 27 patients in his practice felt better during pregnancy. Dr. Klimas suggests that improvement may be due to increased blood volume during pregnancy or hormonal changes.[7]
Postpartum[edit | edit source]
In one survey, after delivery, 30% had no change in symptoms, 20% improved, and 20% got worse. Dr. Klimas reports that her patients typically do well postpartum until about 3 to 6 months after, at which time there is often a severe relapse.[7] Another study indicates that postpartum relapse or worsening of symptoms is likely due to the extra effort of taking care of a baby, as well as the shift away from pregnancy-related hormones and the TH2 state.[9]
Postpartum depression is two to three times more common in mothers with ME/CFS compared to healthy mothers.[9]
Complications[edit | edit source]
When comparing these women's pregnancies after illness onset to pregnancies before illness onset (but not to healthy controls), the rate of complications were similar. In pregnancies occurring after illness onset, there was a higher rate of miscarriages (30% vs. 8%) and development delays or learning disabilities (21% vs. 8%).[8] However, this may be explained by maternal age (pregnancies before illness onset occurred when women were younger than pregnancies occurring after illness onset).[8]
Significant studies[edit | edit source]
Schacterle and Komaroff (2004)[edit | edit source]
Schacterle and Komaroff (2004) conducted a retrospective study on women with ME/CFS who had undergone pregnancy before and/or after onset of illness. Many women with ME/CFS have to make the difficult decision of whether to have a child, and concern due to illness has resulted in lower rates of pregnancy. Of interest, "women who had children before the onset of CFS and decided to continue to have children had more pregnancies after the onset. Among the 19 patients (22%) who reported pregnancies both before and after the onset of CFS, 30 pregnancies occurred before and 46 occurred after the onset."
The study showed that during pregnancy, symptoms were unchanged or improved in 71% of women with ME/CFS. After pregnancy, symptoms were unchanged or improved in 50% of patients. Many women report concern over adverse outcomes for their children, yet there is little evidence that this occurs. Importantly, most maternal and offspring outcomes from pregnancies that occurred after illness onset were not systematically worse than pregnancies occurring prior to illness onset. These data are overall encouraging for women with ME/CFS considering pregnancy. [8]
Pregnancy in other conditions[edit | edit source]
In general, Th1 dominant immune disorders tend to improve during pregnancy while Th2 dominant immune disorders tend to worsen.[10] For example, in a study of women with rheumatoid arthritis 75% of patients experienced remission of their RA during pregnancy and 62% experienced a worsening of symptoms after delivery.[11] 92% of pregnant women with RA relapse within the first three months after delivery.[12] The course of Crohn's disease and ulcerative colitis improves during and after pregnancy.[13] In relapsing-remitting multiple sclerosis, rates of relapse decrease during the first two trimesters and increase significantly postpartum.[14]
By contrast, pregnancy increases rates of lupus flares.[15] In a retrospective study of fibromyalgia patients based on personal interviews, nearly all patients surveyed experienced a worsening of symptoms during pregnancy, especially during the third trimester. Fibromyalgia did not appear to have an adverse effect on the outcome of pregnancy or the health of the baby.[16]
Related potential therapy[edit | edit source]
Human Placental Extract[edit | edit source]
HPE is used as a type of Complementary and Alternative Medicine therapy in various conditions involving chronic inflammation for its anti-inflammatory properties. In one interventional trial, injections of subcutaneous human placental extract (HPE) were found to improve symptoms in ME/CFS patients. Of note, the study did not find a difference between HPE injections and placebo. [17]
See also[edit | edit source]
Learn more[edit | edit source]
- Pregnancy, Motherhood, Parenthood and ME/CFS[18]
- 2010, Pregnancy in Women with Chronic Fatigue Syndrome (ME/CFS)[9]
- 2016, Pregnancy and ME/CFS[19]
- Pregnancy and Chronic Fatigue Syndrome-- Four Mothers Who've Been Through It Talk
References[edit | edit source]
- ↑ "Do pregnancy-related changes in the microbiome stimulate innate immunity?". Trends in Molecular Medicine. 19 (8): 454–459. August 1, 2013. doi:10.1016/j.molmed.2013.06.002. ISSN 1471-4914.
- ↑ Jump up to: 2.0 2.1 "What is the placenta?". Pregnancy, Birth & Baby. July 2017. Retrieved November 14, 2018.
- ↑ Sakamoto, Rie; Osada, Hisao; Iitsuka, Yoshinori; Masuda, Kentarou; Kaku, Kenshi; Seki, Katsuyoshi; Sekiya, Souei (May 2003). "Profile of neurokinin B concentrations in maternal and cord blood in normal pregnancy". Clinical Endocrinology. 58 (5): 597–600. ISSN 0300-0664. PMID 12699441.
- ↑ Jump up to: 4.0 4.1 4.2 Reinhard, G.; Noll, A.; Schlebusch, H.; Mallmann, P.; Ruecker, A.V. (April 28, 1998). "Shifts in the TH1/TH2 balance during human pregnancy correlate with apoptotic changes". Biochemical and Biophysical Research Communications. 245 (3): 933–938. doi:10.1006/bbrc.1998.8549. ISSN 0006-291X. PMID 9588218.
- ↑ Kidd, Parris (August 2003). "Th1/Th2 balance: the hypothesis, its limitations, and implications for health and disease". Alternative Medicine Review: A Journal of Clinical Therapeutic. 8 (3): 223–246. ISSN 1089-5159. PMID 12946237.
- ↑ Munoz-Suano, Alba; Hamilton, Alexander B.; Betz, Alexander G. (May 2011). "Gimme shelter: the immune system during pregnancy". Immunological Reviews. 241 (1): 20–38. doi:10.1111/j.1600-065X.2011.01002.x. ISSN 1600-065X. PMID 21488887.
- ↑ Jump up to: 7.0 7.1 7.2 Allen, Peggy Rosati (July 2008). "Chronic fatigue syndrome: implications for women and their health care providers during the childbearing years". Journal of Midwifery & Women's Health. 53 (4): 289–301, quiz 399. doi:10.1016/j.jmwh.2007.12.001. ISSN 1542-2011. PMID 18586181.
- ↑ Jump up to: 8.0 8.1 8.2 8.3 Schacterle, Richard S.; Komaroff, Anthony L. (February 23, 2004). "A Comparison of Pregnancies That Occur Before and After the Onset of Chronic Fatigue Syndrome". Archives of Internal Medicine. 164 (4): 401. doi:10.1001/archinte.164.4.401. ISSN 0003-9926.
- ↑ Jump up to: 9.0 9.1 9.2 Underhill, Rosemary. "Pregnancy in Women with Chronic Fatigue Syndrome (ME/CFS)" (PDF). NJCFSA. New Jersey Chronic Fatigue Syndrome Association.
- ↑ ØSTENSEN, MONICA (June 1, 1999). "Sex Hormones and Pregnancy in Rheumatoid Arthritis and Systemic Lupus Erythematosus". Annals of the New York Academy of Sciences. 876 (1). doi:10.1111/j.1749-6632.1999.tb07630.x/abstract. ISSN 1749-6632.
- ↑ Østensen, Monika; Aune, Berit; Husby, Gunnar (January 1983). "Effect of Pregnancy and Hormonal Changes on the Activity of Rheumatoid Arthritis". Scandinavian Journal of Rheumatology. 12 (2): 69–72. doi:10.3109/03009748309102886. ISSN 0300-9742.
- ↑ "The impact of pregnancy on rheumatoid arthritis outcome: The role of maternofetal HLA class II disparity". Joint Bone Spine. 77 (1): 36–40. January 1, 2010. doi:10.1016/j.jbspin.2009.11.009. ISSN 1297-319X.
- ↑ "Do pregnancy-related changes in the microbiome stimulate innate immunity?". Trends in Molecular Medicine. 19 (8): 454–459. August 1, 2013. doi:10.1016/j.molmed.2013.06.002. ISSN 1471-4914.
- ↑ Confavreux, Christian; Hutchinson, Michael; Hours, Martine Marie; Cortinovis-Tourniaire, Patricia; Moreau, Thibault (July 30, 1998). "Rate of Pregnancy-Related Relapse in Multiple Sclerosis". New England Journal of Medicine. 339 (5): 285–291. doi:10.1056/nejm199807303390501. ISSN 0028-4793.
- ↑ Ruiz-Irastorza, G.; Lima, F.; Alves, J.; Khamashta, M.A.; Simpson, J.; Hughes, G. R.V.; Buchanan, N.M.M. (February 1, 1996). "INCREASED RATE OF LUPUS FLARE DURING PREGNANCY AND THE PUERPERIUM: A PROSPECTIVE STUDY OF 78 PREGNANCIES". Rheumatology. 35 (2): 133–138. doi:10.1093/rheumatology/35.2.133. ISSN 1462-0324.
- ↑ Østensen, Monika; Rugelsjoen, Anne; Wigers, Sigrid Horven (January 1997). "The Effect of Reproductive Events and Alterations of Sex Hormone Levels on the Symptoms of Fibromyalgia". Scandinavian Journal of Rheumatology. 26 (5): 355–360. doi:10.3109/03009749709065698. ISSN 0300-9742.
- ↑ Park, Sat Byul; Kim, Kyu-Nam; Sung, Eunju; Lee, Suk Young; Shin, Ho Cheol (2016). "Human Placental Extract as a Subcutaneous Injection Is Effective in Chronic Fatigue Syndrome: A Multi-Center, Double-Blind, Randomized, Placebo-Controlled Study". Biological & Pharmaceutical Bulletin. 39 (5): 674–679. doi:10.1248/bpb.b15-00623. ISSN 0918-6158.
- ↑ "Pregnancy, Motherhood, Parenthood and ME/CFS - Emerge Australia". Emerge Australia. Retrieved August 11, 2018.
- ↑ Reynolds, Leigh (July 6, 2016). "Pregnancy and ME/CFS". Bateman Horne Center. Bateman Horne Center. Retrieved August 11, 2018.

